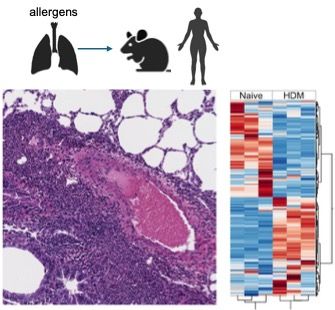
One of the scientific aims of the Gazzinelli Lab is to investigate the upstream immunological events that determine how Th2 effector cells drive type-2 associated pulmonary inflammation using experimental mouse models and human clinical samples. Preliminary transcriptome analyses of House Dust Mite (HDM)-allergic mouse lungs revealed distinct mRNA signature driven by allergens when compared with naïve lungs, profiles that show marked upregulation in signature genes associated with the Th2 activation pathways including IL-4, IL-5, IL-9, IL-13, IL-33 and other important inflammatory mediators, including co-stimulatory molecules on T cells associated with lung response. Indeed, one of the co-stimulatory molecules highly upregulated in effector memory CD4+ T cells of HDM-sensitized lungs is the TNFSF8 also known as CD153 or CD30L. We then decided to investigate the role of CD30L in the differentiation of allergen-specific Th2 cells in the lung tissue of HDM-sensitized mice. Our preliminary data demonstrated that the HDM-sensitization in the CD30L deficient mice failed to induce an allergen-specific memory CD4+ Th2 cell response in the lungs when compared with WT mice, indicated by the marked reduction in the frequency of allergen-specific IL-13 producing memory CD4 T cells, after in vitro stimulation with HDM extract. Most interestingly, the impairment in the development of an effector Th2 response in the CD30L deficient mice led to diminished HDM-specific IgE levels, as well as eosinophils numbers in the lung tissue, a finding that suggests a role for CD30L in the pathogenesis of allergic responses in the tissue. Our next step is to investigate whether anti-CD30L neutralizing antibodies during the effector phase of the HDM-sensitization model in mice could prevent the establishment of a pathogenic type 2 allergic response in the lungs.